

Professor Steve Parker
Professor in Chemistry at The University of Bath
My research interest , summarised below, is to develop and to apply computer simulation techniques to model the structure and reactivity of solids and their surfaces at the atomic scale.
Education: 1969 - 1976 Tiverton Grammar School spot me from
73? (ref)
1976 - 1982 University College London
University of Bath,
Claverton Down,
Bath BA2 7AY,
UK.
Phone: +44 (0) 1225 386505
FAX: +44 (0) 1225 386231
Email: S.C.Parker@bath.ac.uk
SUMMARY OF RESEARCH
The focus of our research is in developing and applying novel computer simulation techniques to extend our understanding of the structure, stability and reactivity of ceramics and minerals at the atomic level. There are broadly three main areas where we apply these techniques
Computer Simulation of Surfaces
Many of the important material properties are controlled by the surfaces. Thus to understand and modify these material properties it is essential to understand the surfaces at the atomic level, which can be achieved with simulation. However, an outstanding major challenge remains, which is the successful treatment of solid surfaces in contact with water. An understanding of the solid-water interface will enable us to investigate factors directly relevant to a wide range of important applications. These range from the effectiveness of minerals for immobilising heavy metals and organic pollutants which need to be removed from the environment to the development of new additives for assisting mineral separation. At a more fundamental level computational techniques can make significant progress in developing an understanding of growth processes at the atomic level and in particular identify the impact of the surface topology, surface impurities and the fluid phase on the kinetics of crystal growth and dissolution.
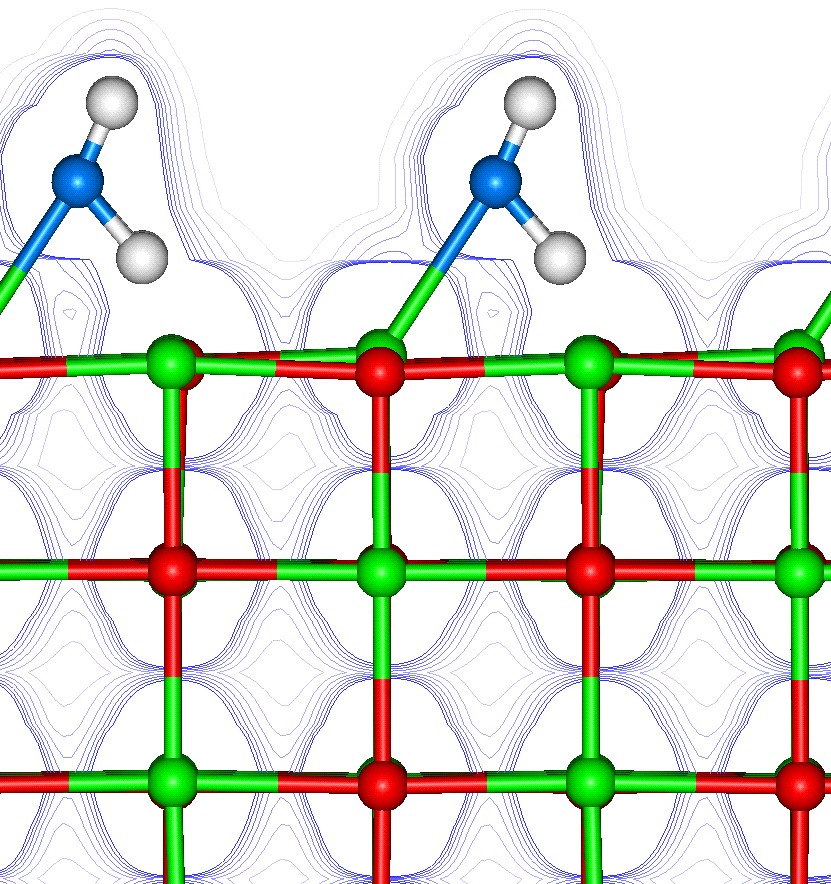
Predictive Modelling of the Structure and Reactivity of Catalysts
The objective of this work is to model chemical reactions occurring at the atomic scale for two classes of material, zeolites, which are microporous solids, and oxide supported catalysts. We aim to develop and apply a range of atomistic and electronic structure techniques. The atomistic simulations are used to characterise the catalyst, by identifying the structure, the effect of defects and the influence of impurities. Target systems include examining Octane adsorbed in the zeolite, MFI catalysts that can remove NOx, which is of environmental interest. It has received much experimental attention but there are still a number of unresolved questions concerning the mechanism.
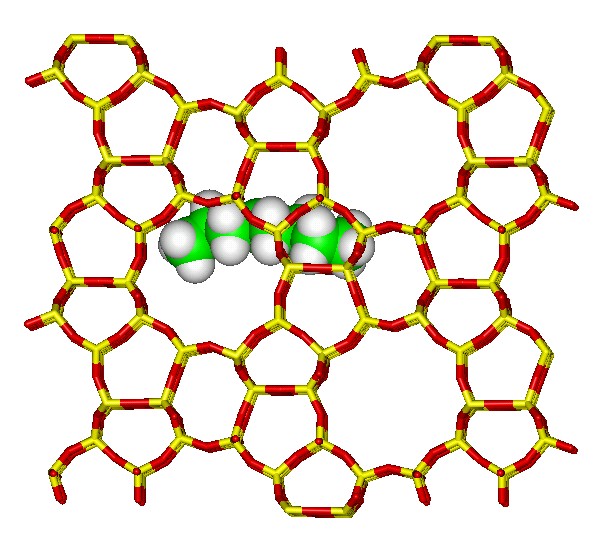
Octane adsorbed in the zeolite
Computer Simulation of Ceramic and Mineral Crystal Structures
In the final area we examine the factors controlling which crystal structure a ceramic or mineral will adopt and the underlying thermodynamics controlling its stability. In addition to its relevance to basic science, this work relates to problems in applied areas such as sensors, coatings, ceramics and mineralogy. Similarly there are many cases in mineralogy and geochemistry where a phase transition is modified by impurities, but often there is the additional concern of the effect of high pressures and high temperatures. For example, the Earth's mantle, which spans a temperature range of up to 3000K and 135GPa.
For further details see the computational solid state group's home page and/or the latest publications
Thanks to Dino 2004

